CAR-T Cellular Therapy Development
CAR-T Cellular Therapy Solutions

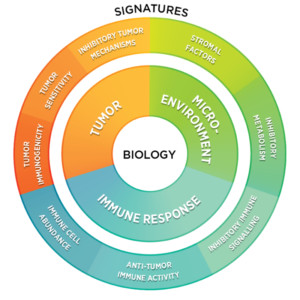
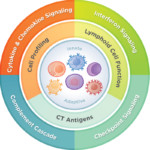
From optimizing CAR-T cell design and evaluating pre-clinical models to discovering response biomarkers and mechanisms of action, together we can advance the field of cellular therapy and improve the human condition.
Featured Solutions
Publications
Two Cases of Severe Pulmonary Toxicity from Highly Active Mesothelin-Directed CAR T Cells
Multiple clinical studies have treated mesothelin (MSLN)-positive solid tumors by administering MSLN-directed chimeric antigen receptor (CAR) T cells. Although these products are generally safe, efficacy is limited.
Biological and Clinical Implications of Gene-Expression Profiling in Diffuse Large B-Cell Lymphoma: A Proposal for a Targeted BLYM-777 Consortium Panel as Part of a Multilayered Analytical Approach
Simple Summary: This review summarizes gene-expression profiling insights into the background and origination of diffuse large B-cell lymphomas (DLBCL). To further unravel the molecular biology of these lymphomas, a consortium panel called BLYM-777 was designed including genes important for subtype classifications, genetic pathways, tumor-microenvironment, immune response and resistance to targeted therapies.
Molecular and Functional Signatures Associated with CAR T Cell Exhaustion and Impaired Clinical Response in Patients with B Cell Malignancies
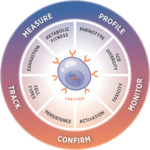
Despite the high rates of complete remission following chimeric antigen receptor (CAR) T cell therapy, its full capacity is currently limited by the generation of dysfunctional CAR T cells. Senescent or exhausted CAR T cells possess poor targeting and effector functions, as well as impaired cell proliferation and persistence in vivo.

Contact Us
Have questions or simply want to learn more?
Contact our helpful experts and we’ll be in touch soon.