The Coronavirus mRNA Vaccine Ushers in Hope for 2021
Vaccines: biological agents eliciting an immune response to a specific antigen derived from an infectious disease-causing pathogen.
The concept of a “vaccine” emerged in 1796 when Edward Jenner developed the first by subcutaneously inoculating the smallpox virus into non-immune individuals.
Vaccines are usually made from inactivated or attenuated pathogens and proteins or polysaccharides called adjuvants known to provoke an immune response. These preparations stimulate the body’s immune system to recognize the foreign agent as a threat, mount an effective immune response, and learn to recognize and disrupt the real pathogen. These traditional approaches to vaccines have been effective in eradicating fatal illnesses such as polio or smallpox but have limitations, either because some pathogens are dangerous even in their inactivated forms, or because the immune system fails to mount an effective immune response, as the fight against HIV has taught us.
Fast forward to 2020 and the coronavirus pandemic; the combined burden of illnesses, deaths and economic & social devastation from COVID-19 has rocked the world. Scientists have been racing to develop a safe and effective vaccine to control the spread of SARS-CoV-2 and bring our lives back to normal.
Using mRNA as a therapeutic molecule is not a new idea: Malone et al. published in 1989 a seminal work in PNAS on a method for RNA transfection, using a synthetic cationic lipid, N-[1-(2,3-dioleyloxy)propyl]-N,N,N-trimethylammonium chloride (DOTMA), incorporated into a liposome.
What are mRNA Vaccines?
Nucleic acid-based vaccines mimic a viral infection to express vaccine antigens in situ, eliciting both cytotoxic T cell and humoral responses, a critical factor for eliminating infections where cellular and humoral immune responses are necessary for efficacy. In addition, they have intrinsic adjuvant properties as they carry specific pattern recognition receptors, critical for maturing dendritic cells to enhance the induction of the adaptive immune response.
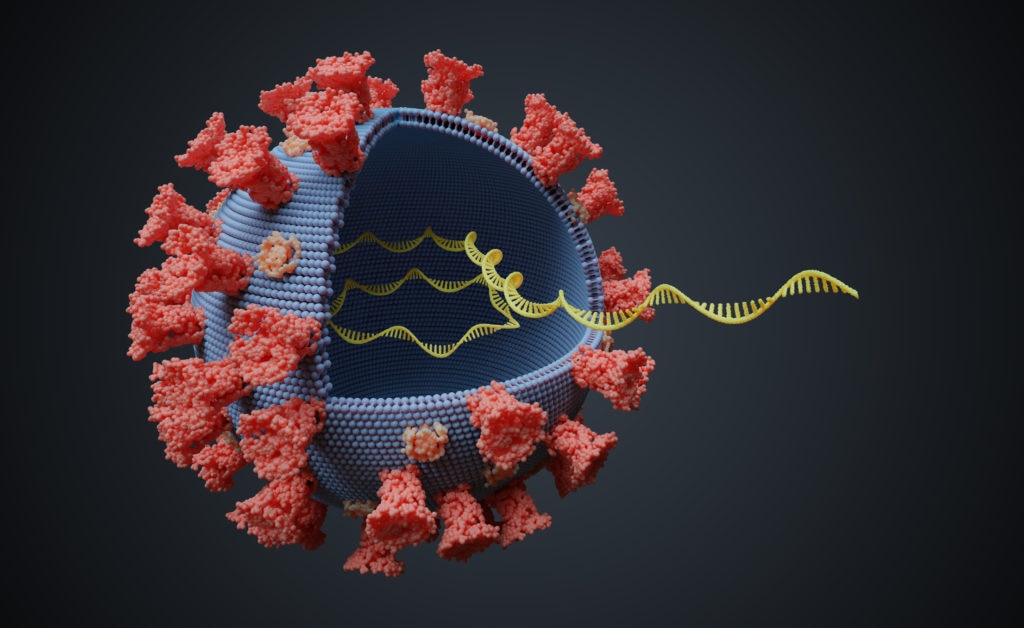
Among nucleic acid-based vaccines, mRNA-based ones have several advantages. They are delivered without the need to cross the nuclear membrane barrier for protein expression and can express complex antigens without packaging constraints. Furthermore – and this will be critical in saving lives – they can be produced rapidly and at a lower cost.
On December 10, 2020, the New England Journal of Medicine finally published a complete, peer-reviewed study describing Phase 2 and 3 clinical trials of a vaccine against SARS-CoV-2. A two-dose regimen of this RNA vaccine produced jointly by BioNtech and Pfizer was deemed safe and 95% effective against the coronavirus, meeting the efficacy endpoints.
The first dose elicited a partial response in a population of over 43,000 subjects. The vaccine was approved in the United States on December 11, 2020, when the FDA issued the first emergency use authorization for a vaccine for the prevention of COVID-19 in individuals 16 years of age and older, allowing for its distribution in the US.
The success of this vaccine in safely preventing COVID-19 is critical from multiple standpoints. The clinical trial showed that COVID-19 could be prevented by effective immunization. Furthermore, this study established the feasibility of RNA-based vaccines as a new, reliable, and quick to mass-produce approach for protection against infectious diseases.
The development of this vaccine was initiated on January 10, 2020, quickly following the release of the viral RNA sequence – as the mRNA nature of the vaccine only requires knowing the pathogen’s genetic information. A similar approach can be used for future pathogens, accelerating the development of a vaccine.
Key to success: Trust and Collaboration between Scientists and the Public.
A more important lesson we have learned from the approval of this first SARS-CoV-2 mRNA vaccine is that the combined power of substantial R&D investment, open & free collaboration among researchers and trust between scientists and the general population is key to driving innovation.
Remember March 2020 when most of the country initially went into lockdown? We at NanoString certainly do. It feels like a century has passed since then. We came together as a company and decided to be at the forefront of finding a solution to the pandemic. In May 2020, we launched the Coronavirus Panel Plus, to be added as a spike-in to nCounter Gene Expression panels and/or Custom CodeSets and the nCounter Host Response Panel, a 785-plex assay containing probes for genes involved in the host response to pathogens.
Last but definitely not least, the GeoMx DSP Protein and RNA assays allowed scientists to visualize the interaction of SARS-CoV-2 with the immune system, the effect of COVID-19 on organ function, and the subsequent contribution of a dysfunctional immune response, as demonstrated by Niyati Desai and colleagues in a recent work published on Nature Communications.
We know that NanoString is not the only company heavily invested in these efforts to fight the COVID-19 pandemic. There was never a more accurate time to say this aloud: we are all in this together!
For Research Use Only. Not for use in diagnostic procedures
References:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1200696/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2786381/