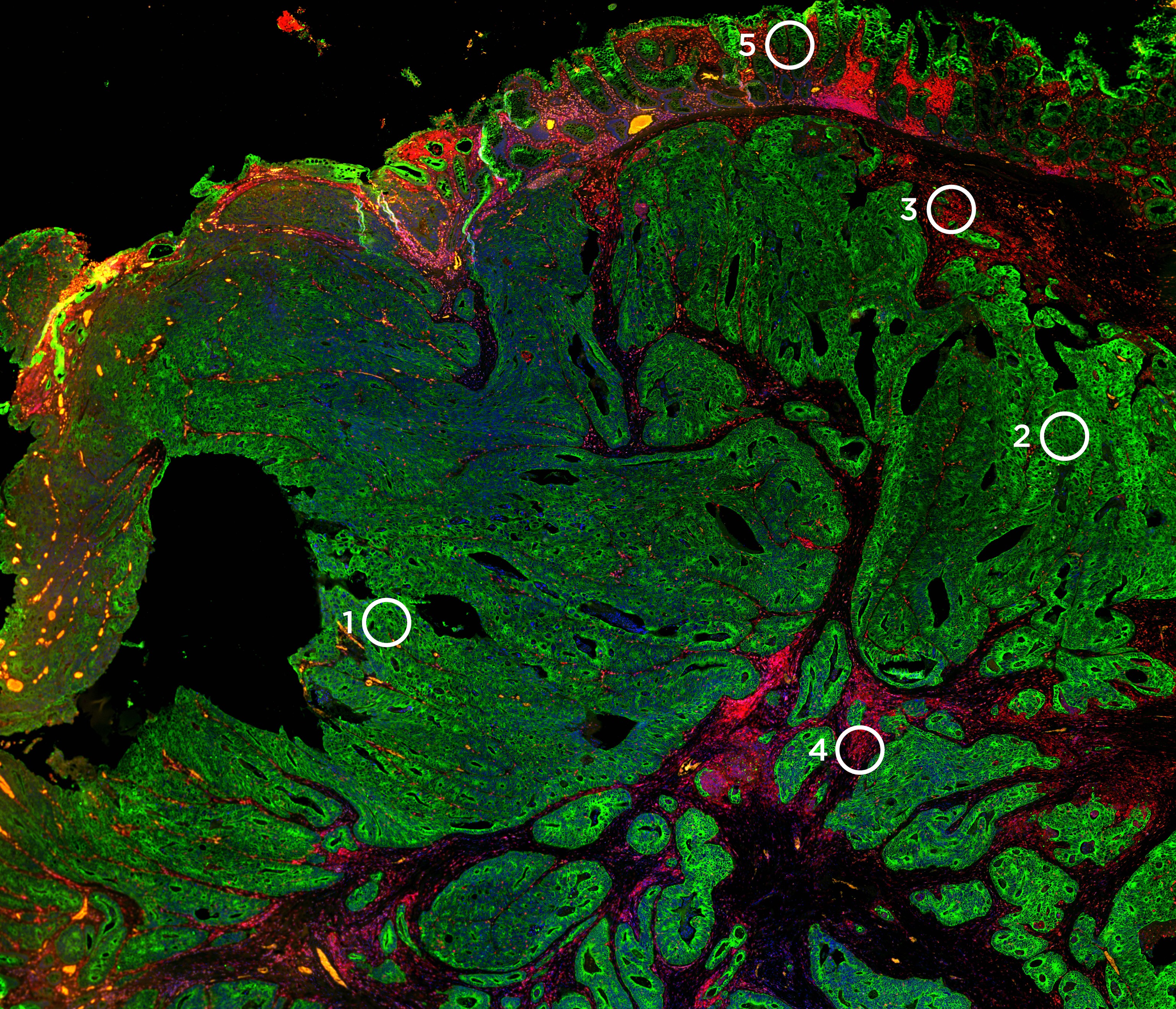
Q&A with Dr. Aubrey Thompson, Ph.D.: The GeoMx® Digital Spatial Profiler is Turning the Tables on Breast Cancer
E. Aubrey Thompson, Ph.D is a Professor in the Department of Cancer Biology at the Mayo Clinic in Jacksonville, FL. His research focuses on breast cancer and using genomic and proteomic tools to identify novel biomarker signatures and new targets for therapies. His center was one of the first to receive the GeoMx Digital Spatial Profiler (DSP). We caught up with him on a sunny California afternoon and asked him to share his experience with the nCounter® Breast Cancer 360™ Panel and GeoMx DSP.

NS: Your academic background is in chemistry and biophysics. What led you to study the genomic architecture of breast cancer?
AT: We were studying an enzymatic mechanism involved in estrogen biosynthesis. I got interested in steroid metabolism, estrogen action, and, more generally, in hormone action. A very large part of my career has been studying how hormones control gene expression within the context of cell proliferation. As global transcriptomic technology became more available, we had the opportunity to look beyond the way hormones regulate individual genes and look at the way hormones regulate the entire transcriptome of the cell. I moved to the Mayo Clinic in 2003 specifically to reinvent myself as a translational researcher. Prior to that, I had been a hardcore basic scientist. I had spent my life studying gene structure and function in cultured cells and I wanted to find out whether anything we had learned was applicable to human physiology and disease.
NS: What do you think is the major roadblock to cure breast cancer once and for all?
AT: Early diagnosis. With the more common kind of breast cancers (human estrogen receptor positive breast cancer) the cure rates for Stage 1 disease are 99%. There’s not much scope for improving that cure rate. There are, however, a number of subtypes that are more aggressive. They are sometimes more difficult to diagnose, tend to be diagnosed at a later stage, tend to be diagnosed in younger women, and the outcome for these cancers is significantly worse. The challenge for us is to identify patients who are not going to do well even with the excellent therapeutic interventions we have now so that we can focus on them.
NS: What are your thoughts on the increasing use of biomarkers in cancer treatment?
AT: Biomarkers are critical. There are so many new biomarkers coming up and some of the therapeutics work very well for the general population but won’t work well on the other subset of patients. So, identifying that subset is key and it’s not really different from individualized/personalized medicine. It’s using genomic and proteomic technology to match the right treatment to the right patient. That’s where I think the NanoString technology has so much scope; a lot of the NanoString platforms have real potential for matching the right patient with the right type of treatment.
NS: Why is immunotherapy less impactful in breast cancer when compared to other kinds of cancer?
AT: I think there’s more than one answer to that question. In order to answer it, we have to understand more about the immunobiology of breast cancer. Up to this point, a lot of the studies have focused on what is going on in the stroma (the supportive and connective tissue around the breast). I think that the stromal tissue immunobiology is going to be less important than the biology of the immune cells that are interacting directly with the tumor. This is where the GeoMx DSP system opens up a whole new landscape for us.
NS: Did you discover an unexpected relationship between breast cancer cells and the tumor microenvironment?
AT: Yes! We thought the stroma was going to be much more important and that we were going to learn a lot more than we have so far. The fact that we’re seeing so much more in the tumor is, in and of itself, unexpected.
NS: What about the GeoMx DSP makes it an ideal platform for your research?
AT: It’s a simple answer- the ability to look at numbers, types, activities, and locations of immune cells. It enables us to look at a great many more immune features, such as the abundance of an immune protein in a particular location, and to correlate those features with the outcome. We can then learn more about the basic biology of breast cancer and apply that knowledge to understanding how therapeutics work. The GeoMx DSP is game-changing.
NS: How does the nCounter Analysis System and GeoMx DSP together make your work easier as compared to other transcriptomic and proteomic platforms?
AT: We’ve used the classical approach, beginning our studies with high-throughput, high-depth sequencing. We look at large numbers of samples and develop hypotheses that we refine using NanoString. NanoString is ideal for screening signatures and pathways which are more challenging to do when you are simply using sequencing data because that classic approach requires a lot of additional analytical steps. Those steps are built into the NanoString process. The greatest advantage we have gained by using NanoString is the ability to analyze clinical samples. The vast majority of clinical samples that we have access to are FFPE samples (formalin-fixed, paraffin-embedded). They’re not very good for RNA sequencing but they are excellent for the nCounter and GeoMx DSP. The ability to use RNA extracted from FFPE samples to measure gene signatures and pathway signatures is very important to us.
NS: How did the Breast Cancer 360 (BC 360) panel impact your work?
AT: We just published a paper using the BC 360 panel to identify a subset of triple negative breast cancer patients who benefit from fluoropyrimidine adjuvant therapy. I think the GeoMx DSP is going to figure more prominently into our work. We’re beginning to learn more at intrinsic subtypes within particular breast cancer subtypes. We look for HER2 positive breast cancer samples and within that group we identify subsets that have either basal-like or luminal-like features. Very often, those two subtypes have different therapeutic profiles. The intrinsic subtype signatures are going to be very useful going forward but that hinges on the ability to identify them.
NS: How will the data from the BC 360 panel inform your future GeoMx DSP experiments?
AT: I think the data will inform our conclusions. We want to be able to integrate RNA data with proteome data. There are some aspects of tumor biology that we really can’t analyze very well at the protein level. There are some key features of the immune system for which we don’t have good antibodies; those will ultimately have to be measured by RNA. For example, secreted cytokines are difficult to measure at the protein level, but they can be measured by RNA expression. I think you must have both the RNA gene expression data from the BC 360 Panel and the spatial distribution of protein by GeoMx DSP to be successful.
NS: Why is sample conservation so important in your studies?
AT: We have access to large numbers of diagnostic biopsies, or core biopsies; these are equal to the inside diameter of a 14-gauge needle—about 0.6 mm in total diameter. You can’t use all of that to do one experiment—the pathologist simply won’t let us do that. But with GeoMx DSP, we now can take a 5 mm section of that biopsy and measure the abundance of 60 or more proteins in a spatially defined manner. This new technology is unparalleled in terms of its strength to glean information from these small samples. There are millions of these samples available but access to them has been limited in part by the lack of technology that can be used with such small samples. The reality is that these samples are irreplaceable and cannot be exhausted by one or two experiments.
NS: What impact will this have on patients?
AT: The broad answer is that in order to take advantage of immunotherapy in breast cancer, we will need to learn more about its immunobiology. As we learn more about that, we will be able to devise more effective means of immunotherapy and conceivably identify some new targets. I think we’ll also be able to identify subgroups of patients who will or will not benefit from a immunotherapy. I personally think GeoMx DSP will be critical for that. My pathology colleagues are looking at GeoMx DSP as a way to move from traditional scoring methods (1+, 2+, 3+, etc.) to a quantitative scoring method. Today, pathologist evaluate tumor samples for multiple types of receptors (estrogen, progesterone, Ki-67, to name a few). These are all separate assays and they are not quantitative. I think it’s conceivable that DSP can give you all these readings from a single slide, with results measured in quantitative values. I think that specificity of information will have great clinical relevance.
NS: You are likely collaborating with other GeoMx costumers on breast cancer studies. What do you see as the value in pooling DSP data?
AT: I have seen the benefits of collaborations in terms of pooling rare samples and pooling intellectual information. I think that with the GeoMx DSP technology there’s tremendous value in getting users together to talk about both new experiments as well as routine matters, such as best practices in normalizing data. There are a lot of questions that need to be answered and instead of answering them one at a time, let’s get together and talk about how to apply the technology. Another advantage is in sharing data. Typically, none of us have enough samples to answer a question and then validate the answer we have. Collaboration provides an opportunity for validation using samples from other groups.
NS: What should people know about you and your research that we haven’t covered?
AT: I’m motivated to decrease breast cancer mortality because I feel it’s our duty to do so. Our community is very supportive, and I love the people that I work with, both academic colleagues and the scientists at NanoString. When I get up each Monday morning, it’s with enthusiasm and excitement. Mainly, I do what I do because I love it!
FOR RESEACH USE ONLY. Not for use in diagnostic procedures
Links and References
Aubrey Thompson, Ph.D.
https://www.mayo.edu/research/faculty/thompson-e-aubrey-ph-d/bio-00028180
The DONNA Foundation funds ‘game-changing’ research equipment for the Mayo Clinic
Asleh K. et al, Predictive Biomarkers for Adjuvant Capecitabine Benefit in Early-Stage Triple-Negative Breast Cancer in the FinXX Clinical Trial. Clin Cancer Res. 2020 Jan 31.