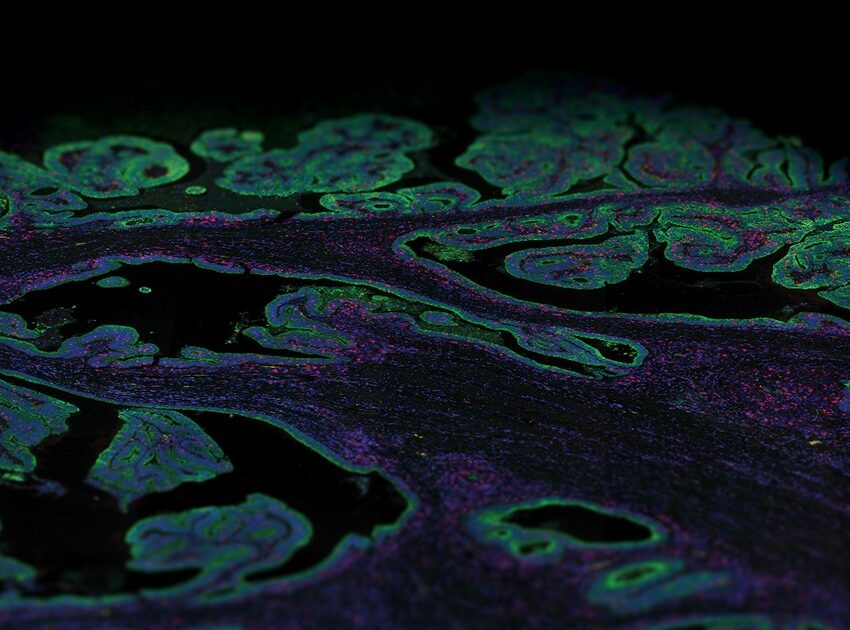
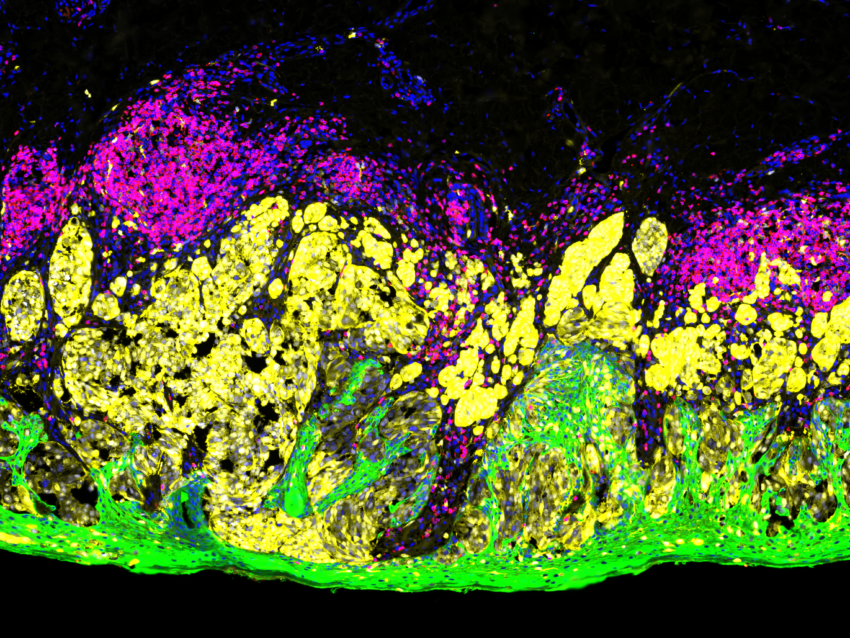
AMP-ed Up for Clinical Evaluation with GeoMx DSP
The Association for Molecular Pathology (AMP) joined the list of conferences to go virtual in 2020. NanoString was online with workshops highlighting uses for the groundbreaking GeoMx® Digital Spatial Profiler (DSP) for pathologists and their studies. The seminar “NanoString GeoMx® Panels: Strategies for Clinical Validation” starts with an educational introduction to the technology itself. Dr. Kit Fuhrman of NanoString leads the viewer through a clear and concise primer on how GeoMx DSP adds another dimension of analysis to traditional pathology techniques. This segment is followed by an informative presentation by Chris Corless, MD, Ph.D., of Oregon Health & Science University. He is Vice-Chair of the Molecular Pathology department and is Chief Medical Officer of Knight Diagnostic Laboratories. Their mission is to “develop and adapt the rapid advances in diagnostic testing that are making personalized medicine a reality.” His talk on research clinical validation strategies and studies is a shining example of realizing that mission.
Dr. Corless has used slide-based pathology assays as part of the SMART program (Serial Measurements of Molecular and Architectural Responses to Treatment). Initially, this program focused on breast cancer and has since expanded to pancreatic cancers and leukemia. The SMART program performs deep analyses on fresh tumor biopsies from patients with advanced metastatic disease. Using the data collected from the profile, physicians then recommend the best relevant therapy for that patient. Patients are monitored, and those with positive progress continue that therapy. Nonresponding patients are re-biopsied and reassessed, creating a real-time approach to identifying and targeting possible tumor vulnerabilities.
Their studies started on slide-based assays to assess the tumor microenvironment using multiplex IHC. This is now being expanded upon by using the GeoMx DSP to focus on intracellular signaling events, starting with NanoString’s Vantage 3D™ Solid Tumor Panel. This approach let them assess therapeutic responses within the tumor and the team focused on phosphorylated markers of signaling proteins. Phosphorylation is a highly labile process that can be lost or distorted by improper or even delayed samples’ fixation. In response to this challenge, the SMART program limits their clinical assays to rapidly fixed samples and has implemented workflows to expedite fixation during biopsies. Subsequent experiments confirmed the preservation of phosphorylation on the markers, paving the way for more in-depth analysis of signaling proteins and incorporation of GeoMx DSP.
A research clinical validation is underway using the GeoMx DSP for a wide-ranging, retrospective analysis of all previously collected biopsies in the SMART biopsy archives. Both the tumor-specific and tumor-adjacent regions are being examined independently, quantitating the data in the previously validated nCounter. This study includes bioinformatic studies and analyses to determine statistical variation between samples from the same patient over time, critical to the SMART therapy goals. As an example, Dr. Corless describes a retrospective study where the progress of a patient with metastatic breast cancer using the slide-based assay. The patient received many different therapies over the course of their treatment. The team compared four different biopsies taken during four different therapies. The biopsy taken at the height of the serum biomarker response to tumor burden is noticeably higher than for a subset of clinically relevant phosphorylated markers when compared to samples taken at different times during other treatments. Although this case study was performed on a slide-based assay using the nCounter, it demonstrates the feasibility of using NanoString technology to examine intracellular signaling events in real-time for patient therapies. SMARTs mission of real-time adaptation to individual patient needs just got a little more real.